with “good manners” during manufacturing (GMP)
In order to ensure the quality of our medicinal products, it is important that measures are taken during production to ensure that the products are manufactured in the prescribed quality.
The requirements for quality assurance of production processes with the aim of process verification are formulated by the European Commission in the principles and guidelines of “Good Manufacturing Practice (GMP)” for medicinal products for human use. For us as a certified company, these “good manners” during production are our top priority.
GMP stands for quality assurance
For years we have been achieving official GMP certification for the production of raw materials and finished products through the responsible and targeted implementation of international specifications. In this manner, we ensure a consistently high quality and safety of the medicines in use. This is also evidenced by the very good tolerability of the products in use over decades.
Thanks to a documented management system that has grown over many years, we ensure the traceability and retraceability of production. An intensive training system also guarantees that every employee at our company works according to the latest specifications. Changes to procedures and methods are carried out by means of an internal control system in compliance with officially deposited documentation.
Trust is good, control is better!
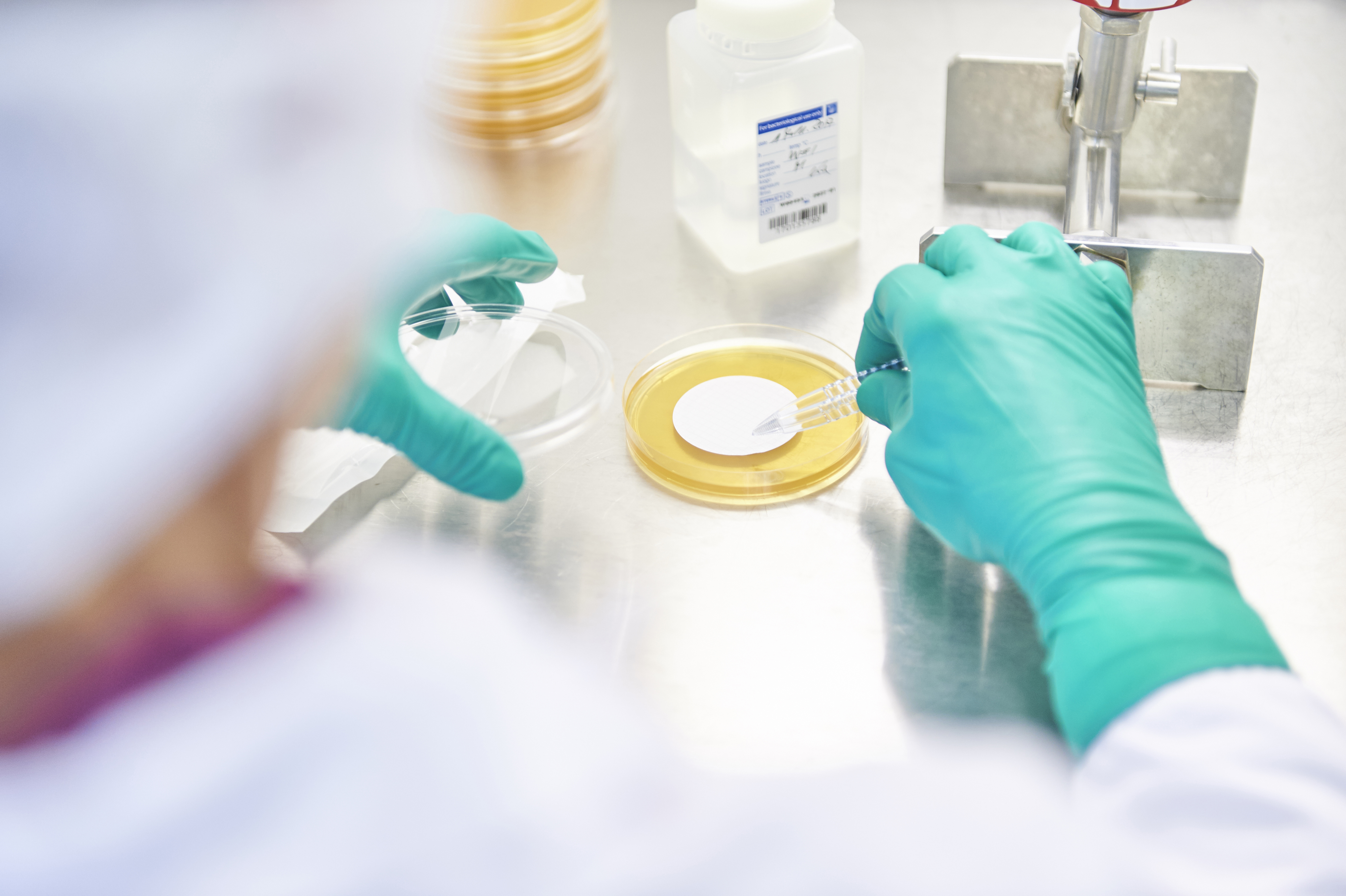
Our in-house quality control department intensively checks all the raw materials and finished products we produce, as well as the auxiliary materials and material we purchase.
National and international standards are adhered to in the process. The examination according to the Homoeopathic Pharmacopoeia (HAB) and the European Pharmacopoeia (Ph. Eur.) is a matter of course for us.
Highest quality of our raw materials
We ensure proof of quality by means of elaborate, validated analysis of the ingredients. We use highly precise and sensitive methods to be able to prove the consistently high quality from batch to batch. One of these methods is capillary electrophoresis, which is used for the majority of our source materials.
The further development and research of the source materials is also a task of our quality control, which it carries out together with renowned laboratories both in Germany and abroad.